In an article appearing online today (preprint available on arXiv) (22nd of December) in the New Journal of Physics, published jointly by the Institute of Physics and Deutsche Physikalische Gesellschaft (DPG), Dr Stéphane Dorbolo and colleagues from University of Liege in Belgium and at the Collège de France have proposed a mechanism that describes the formation and movement of antibubbles through liquid. Antibubbles (figure 1) are
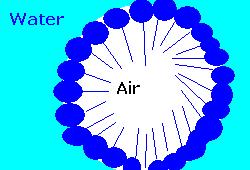
 the exact opposite of bubbles: where bubbles (figure 2) are thin surface of fluid in air surrounding a pocket of air, an antibubble is a thin surface of air in fluid surrounding a pocket of fluid. Although first observed and studied almost a century ago, no one until now has been able to determine how they form.
the exact opposite of bubbles: where bubbles (figure 2) are thin surface of fluid in air surrounding a pocket of air, an antibubble is a thin surface of air in fluid surrounding a pocket of fluid. Although first observed and studied almost a century ago, no one until now has been able to determine how they form. Dr Dorbolo and his coworkers created and filmed antibubbles in a variety of fluids, watching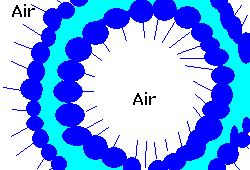 their creation, movement and bursting. Antibubbles are created by pouring a liquid containing a surfactant (figure 3) on to an identical liquid. If the two liquids meet at the right speed, a thin layer of air can be trapped between the two bodies of fluid. As the fluids combine, a part of this film can wrap around a pocket of the fluid, forming an antibubble.
their creation, movement and bursting. Antibubbles are created by pouring a liquid containing a surfactant (figure 3) on to an identical liquid. If the two liquids meet at the right speed, a thin layer of air can be trapped between the two bodies of fluid. As the fluids combine, a part of this film can wrap around a pocket of the fluid, forming an antibubble.
 Dr Dorbolo said "We also found that when they die, or burst, they morph into a form of structure which we have nicknamed the jellyfish form because it looks very like a jellyfish swimming through water. It slowly moves and fades away until it disappears altogether."
Dr Dorbolo said "We also found that when they die, or burst, they morph into a form of structure which we have nicknamed the jellyfish form because it looks very like a jellyfish swimming through water. It slowly moves and fades away until it disappears altogether."
For fun, they also attempted to create antibubbles in an international symbol of Belgium - beer. They initially felt that it would be impossible to form antibubbles - it is impossible to form antibubbles (or bubbles) using pure water, alcohol or oil - a surfactant is needed. The existence of antibubbles implies that there must be a surfactant in the beer. Does this mean that Flemish beer is dishwater? Obviously not (to those of us who've tried it) - the researchers identified proteins common in beer that acts as surfactants.
Dr Dorobolo continued "Antibubbles are mysterious phenomena but we now understand them much better. We have come up with a good model describing how they form and move and have also learnt more about the type of liquids you can create them in. We tried to create them in beer for fun, and didn't think it would be possible, but were amazed when we managed to create giant antibubbles which lasted for almost two minutes and that moved around a glass of beer before bursting."
With care you can create antibubbles in your kitchen.
No comments:
Post a Comment